|
11/24/2023 0 Comments Isotopic carbon
Δ C 13 s a m p l e = ( C 13 / 12 C sample C 13 / 12 C s t a n d a r d − 1 ) ⋅ 1000 Īs a very redox-active element, sulfur can be useful for recording major chemistry-altering events throughout Earth's history, such as marine evaporites which reflect the change in the atmosphere's redox state brought about by the Oxygen Crisis. 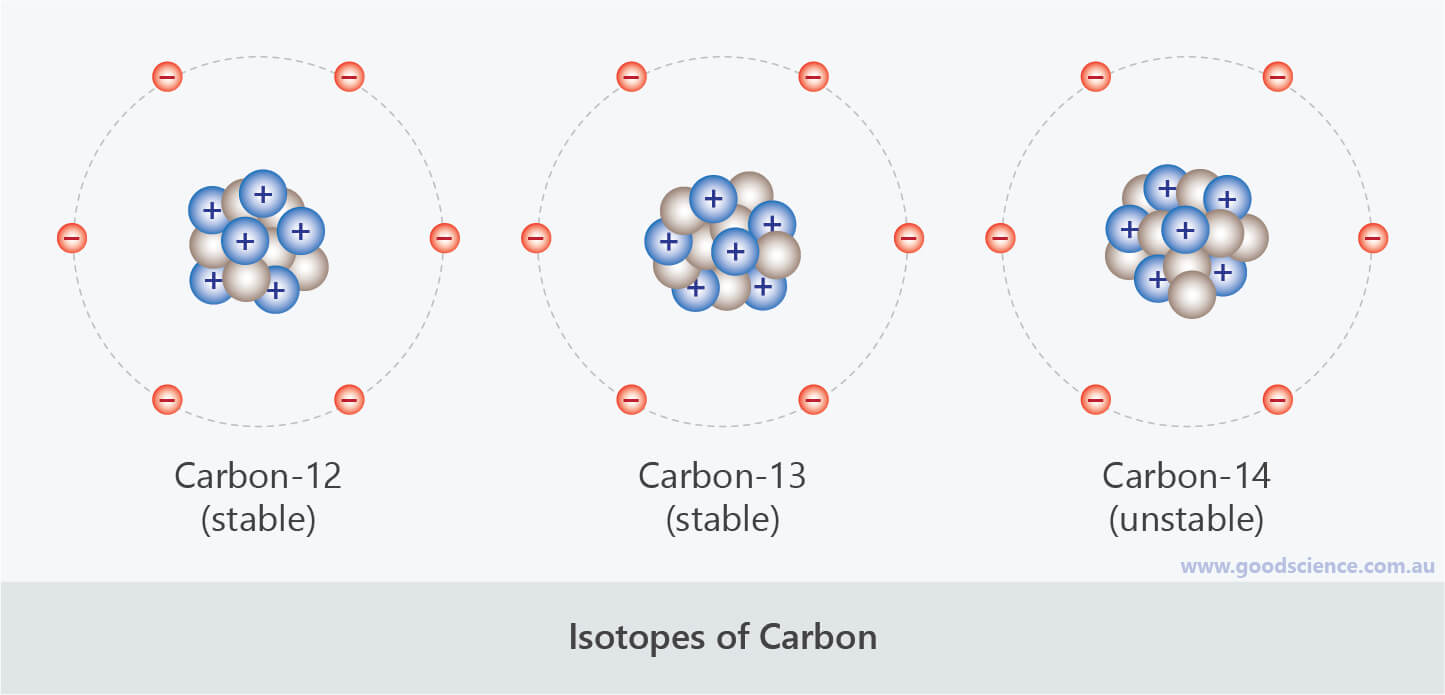
C-13 is used for instance in organic chemistry research, studies into. The ratio is calculated with respect to Pee Dee Belemnite (PDB) standard: Carbon isotopes and mainly C-13 is used extensively in many different applications. The next heaviest carbon isotope, carbon-13 (13 C), has seven neutrons. In geochemistry, paleoclimatology and paleoceanography this ratio is called δ 13C. By far the most common isotope of carbon is carbon-12 (12 C), which contains six neutrons in addition to its six protons. For example, different sources and sinks of methane have different affinity for the 12C and 13C isotopes, which allows distinguishing between different sources by the 13C/ 12C ratio in methane in the air.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
