|
11/22/2023 0 Comments 41 element in periodic table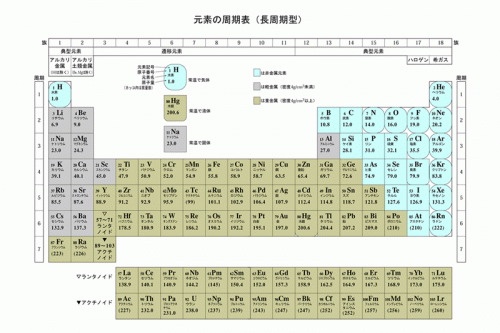
Elements 70 ( Ytterbium Yb) and 71 ( Lutetium Lu) 
This was a compromise of sorts the IUPAC accepted tungsten (element 74) instead of wolfram (in deference to North American usage) and niobium instead of columbium (in deference to European usage). IUPAC officially adopted niobium in 1950 after 100 years of controversy. In the 1860s, it was found that niobium and columbium are the same element and are distinct from tantalum. In 1846, Heinrich Rose discovered that tantalite contained an element similar to tantalum and named it niobium. Elements 41 ( Niobium Nb) and 74 ( Tungsten W) Ĭharles Hatchett named element 41 columbium in 1801 ( Cb), but after the publication of On the Identity of Columbium and Tantalum by William Hyde Wollaston in 1809, the claims of discovery of Hatchett were mistakenly considered refuted. George William Featherstonhaugh, one of the first US geologists, suggested that the element should be named "rionium" after del Río, but this never happened. Later that same year, Friedrich Wöhler confirmed del Río's earlier work. He chose to call the element vanadin in Swedish (which has become vanadium in other languages including German and English) after the Old Norse Vanadís, another name for the Norse Vanr goddess Freyja, whose facets include connections to beauty and fertility, because of the many beautifully colored chemical compounds it produces. In 1831, Sefström of Sweden rediscovered vanadium in a new oxide he found while working with some iron ores. Del Río thought himself to be mistaken and accepted the statement of the French chemist that was also backed by del Río's friend Alexander von Humboldt. The French chemist Hippolyte Victor Collet-Descotils incorrectly declared that del Río's new element was only impure chromium. He later renamed this substance erythronium, since most of the salts turned red when heated. Through experimentation, he found it to form salts with a wide variety of colors, so he named the element panchromium (Greek: all colors). He discovered the element after being sent a sample of "brown lead" ore ( plomo pardo de Zimapán, now named vanadinite). 
Vanadium (named after Vanadís, another name for Freyja, the Scandinavian goddess of fertility) was originally discovered by Andrés Manuel del Río (a Spanish-born Mexican mineralogist) in Mexico City in 1801. In most cases, the controversy was due to a priority dispute as to who first found conclusive evidence for the existence of an element, or as to what evidence was in fact conclusive. However, the names of several elements have been the subject of controversies until IUPAC established an official name. Such an arrangement helps explain the periodicity and periodic trends observed across the elements of the periodic table.The currently accepted names and symbols of the chemical elements are determined by the International Union of Pure and Applied Chemistry (IUPAC), usually following recommendations by the recognized discoverers of each element. The N shell containing 4s, 4d, 4p and 4f, can carry 32 electrons. The M shell contains 3s, 3p, and 3d, and can carry 18 electrons. The K shell contains a 1s subshell hence it can carry 2 electrons, the L shell has 2s and 2p, and can carry 8 electrons. This decides the electron capacity of the shells. 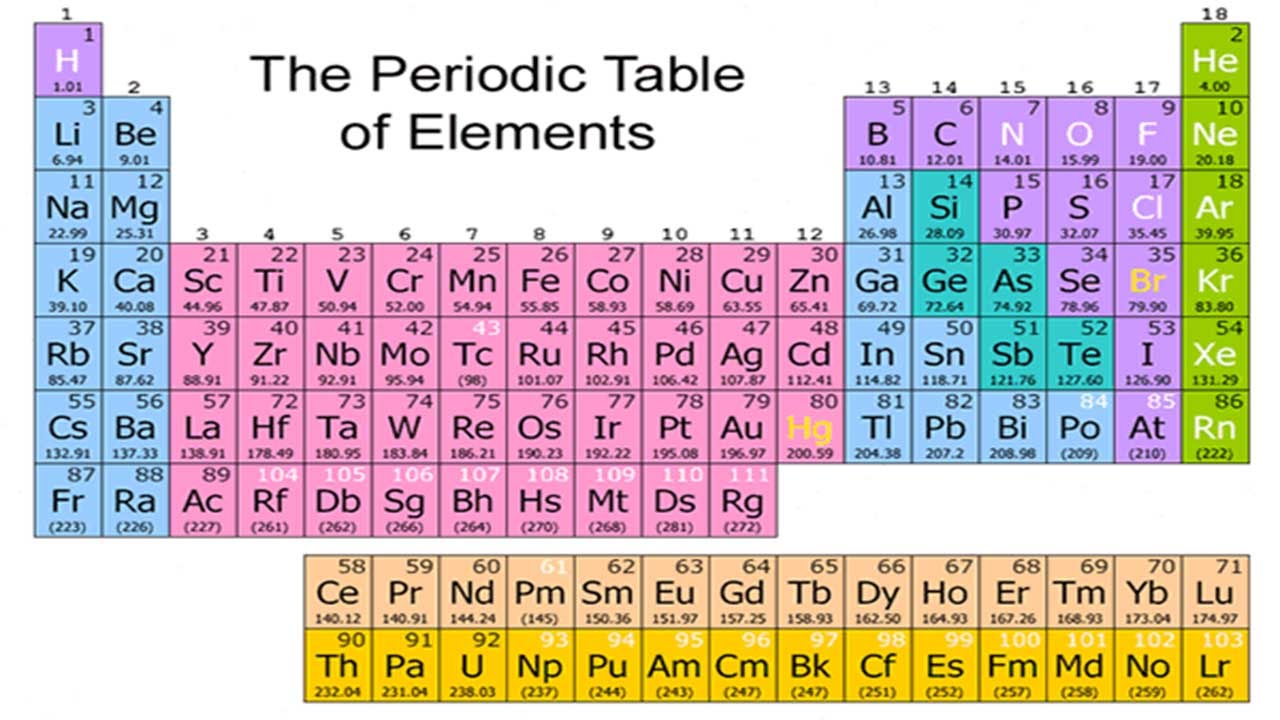
The maximum electrons that can be carried by the sub-shell S is 2, by P is 6, by D is 10, and the F sub-shell can carry 14. Each shell and subshell have a limitation on the amount of electrons that it can carry. The subshells have a distinct shape and configuration, in which the electrons move freely. They stand for sharp (S), principal (P), diffuse (D), and fundamental (F). The shells are labeled K, L, M, N, and so on, from the innermost to the outermost shell.Įach shell has subshells that are named for the type of emission lines produced from different states of angular momentum. This model has been widely accepted, and according to it, each atom has shells, which further have subshells. It involves the specific arrangement of electrons in shells and sub-shells of Bohr’s atomic model. The concept of electronic configuration has replaced the older concept of valency and valence electrons. The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
